 MENU
MENU
Our team consists of professionals in medicine, pharmaceuticals, and software development.
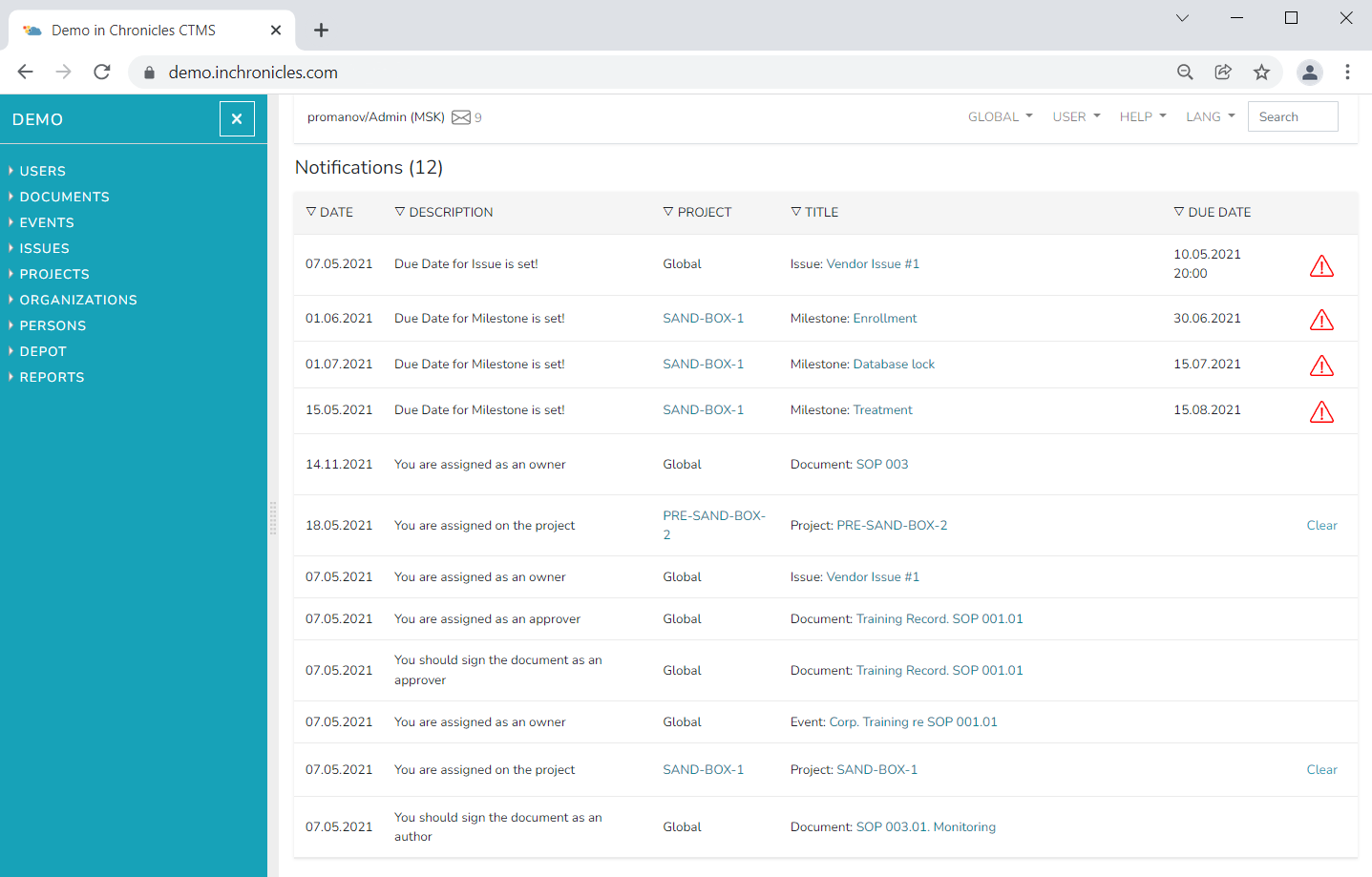
We automate processes for pharmaceutical companies, medical device manufacturers, laboratories, and distributors. Users have efficient tools for process management and monitoring. We test and validate our software before each release, ensuring the reliability of data storage and integrity. Chronicles Research Team is a provider of software products for the GxP industry, making electronic systems accessible for small and medium-sized businesses.
Our products include quality management systems, incident tracking, electronic patient records, and integrated modules for inventory control and supply tracking. We strive to ensure compliance with regulatory requirements and security standards such as GxP and FDA. Our team has a deep understanding of the industry and experience in developing software that addresses the specific needs of medical and pharmaceutical organizations. We take pride in providing innovative and reliable solutions to our clients, helping them increase efficiency, reduce costs, and ensure high data security.

We provide automation services of quality management systems starting from SOPs development and training to systems integration and business processes adapation. Our personnel have expertise in GxP and also have been involved in many research organizations audits and software development. Purchasing the system you get the full package of the documents, training and support. So you can focus on your business and delegate us all the technical issues.

We develop and support systems from the very begining to the ready-to-use product using modern agile methodology and feedback given by clients. Satisfaction of customers needs is our main concern.
We offer flexible terms of the system use. Free trial access allows to test the key functionality. Standard plan with fees per user count and storage usage. We also have special offer for research and educational organizations. Contact us to receive a commercial proposal.